Surgical Management of Intestinal Obstruction from Phytobezoar
Article information
Abstract
Purpose
Phytobezoar is the most common type of bezoar, which can occasionally present as an intestinal obstruction. In this study, the surgical experience and outcome in the management of intestinal obstruction caused by bezoars are described.
Methods
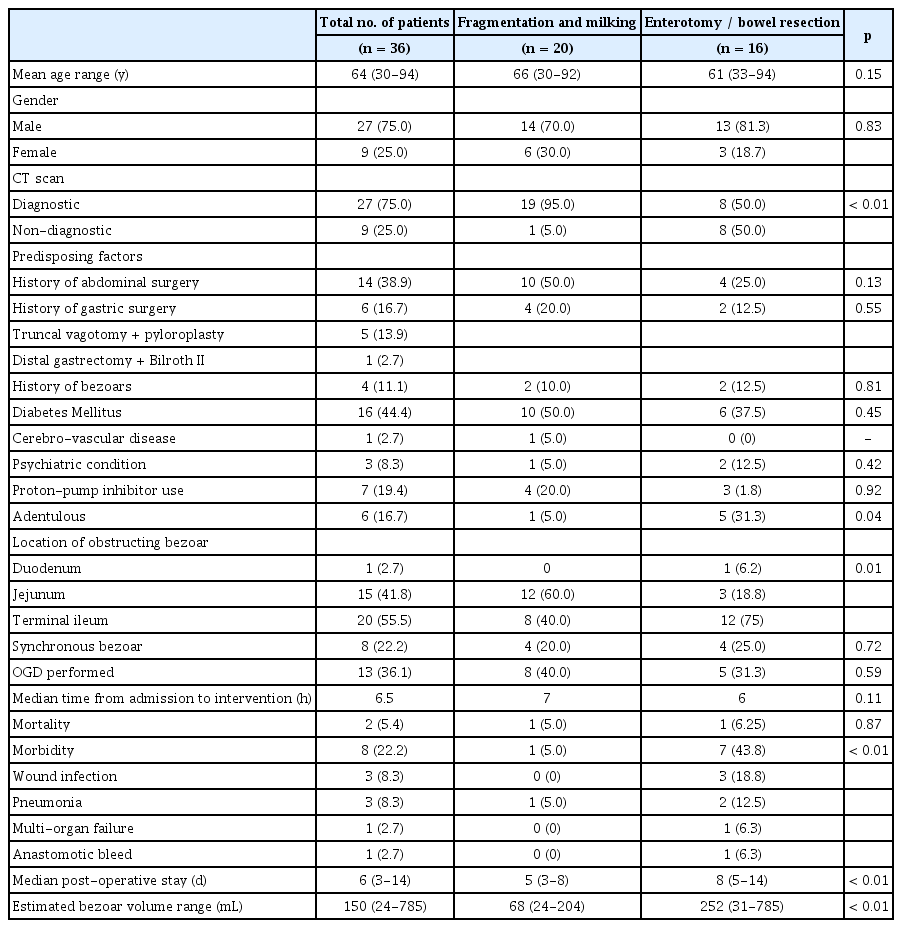
A retrospective analysis of all operative cases of bezoars (n = 36) at Khoo Tech Puat hospital between 2011–2017 was performed. Patient demographics, imaging and operative findings, characteristics of bezoars and related morbidities were analyzed. The study population was subdivided into 2 groups based on operative intervention (fragmentation and milking of bezoars, versus enterotomy and/or bowel resection).
Results
There were 36 cases of bezoars in 35 patients that were included in this study. Computed tomography scans were diagnostic of bezoars in 27 cases (75%). There were 20 cases (55.6%) that underwent fragmentation and milking of bezoars. The remaining 16 cases (44.4%) required an enterotomy or bowel resection. Bezoars that required enterotomy / bowel resection were more likely to be distally located in the ileum (75% vs 40%, p = 0.01), larger in volume (86.5 mL vs 63 mL, p = 0.04), with significant increase in morbidity rates (43.8% vs 5%, p < 0.01) compared with all other cases of bezoars.
Conclusion
Risk factors for enterotomy / bowel resection in bezoar bowel obstruction include, nondiagnostic computed tomography scans, distally located, and larger volumes of bezoars. Fragmentation and milking should be attempted first as it has lower morbidity rates than enterotomy / bowel resection surgery.
Introduction
Bowel obstruction is a common acute presentation in general surgery. Bezoars are defined as aggregates of inedible or undigested material found in the gastrointestinal tract. They account for approximately 4.5% of bowel obstruction cases [1]. Bezoars are mainly classified according to their contents: phytobezoars (vegetable matter), trichobezoars (hair), pharmocobezoars (medications), and lactobezoars (milk proteins). Phytobezoars are the most common type of bezoars, accounting for about 40% of all reported bezoars. There is variation in the incidence of phytobezoars depending on ethnic groups and geographic locations, since phytobezoar formation is related to local food consumption. For example, higher rates of persimmon fruit phytobezoar have been reported in regions with high consumption of orange and persimmon fruits (Asia, Spain, Israel and Turkey) [2-5], and due to the hard consistency, and high tannin content of the fruit, phytobezoars can form [6].
Clinical manifestations vary depending on the location of the bezoar in the body. Bezoars can be discovered incidentally during an endoscopy or imaging, in asymptomatic patients. Bezoars can also be located because of symptoms such as gastric ulceration, or gastric outlet obstruction in the stomach, or as a result of a bowel obstruction (if it is lodged in the small bowel). Previously, it was difficult to clinically differentiate a bezoar-induced small bowel obstruction (SBO), from other causes of SBO such as adhesions because they can present similarly. However, with the availability and the wide-usage of computed tomography (CT) scans, the diagnosis and treatment of SBO from bezoars can be made much quicker [7]. While there are many methods described in the literature to treat bezoars (including dissolution with Coca-Cola or endoscopic retrieval) [8,9], these methods are usually reserved for nonobstructed cases. For patients presenting with acute SBO, surgery is usually performed that includes fragmentation and milking of the bezoar, enterotomy and retrieval, or even bowel resection.
SBO caused by a bezoar is not an uncommon presentation in the Asian population in Singapore. We review the surgical management and outcomes for this group of patients, and present our experiences in this study. We hypothesized that the surgical approach in treating bowel obstruction caused by a bezoar is linked to the size of the bezoar.
Materials and Methods
Hospital records of all patients that underwent surgery for bezoars, over a 6-year period (2011–2017) in Khoo Tech Puat Hospital, Singapore, were retrieved and examined. The electronic operative database was searched using the keyword “bezoar.” All retrieved, operative cases with bezoars found intra-operatively were included. All the operations involved a midline laparotomy. The following data were obtained for analysis: patient demographics, previous medical and surgical history, clinical signs and symptoms, radiological findings, operative findings, morbidities and mortalities. Hospital Institutional Review Board approval was waived and the authors declared no conflict of interest or funding associated with this research.
Patients were sub-divided into 2 groups based on operative intervention. One group required only fragmentation and milking of the bezoars and the other group required enterotomy and/or bowel resection. Statistical analysis was performed using a Statistical Package for the Social Sciences version 20. Assumption of normality was tested using Kolmogotov-Smirnov test and showed that the variables in the study were largely non-parametric. Fisher exact test was used for categorical variables, and Wilcoxon Signed-Ranks test was used for contiguous variables. Statistical significance was reached when p < 0.05.
The sizes of the bezoars were also estimated based on intra-operative measurements, and volumes calculated based on the formula for a cylinder (πr2h) because most bezoars conform to a longitudinal shape in the small bowel.
Results
In this study population, there were 36 cases of SBO resulting from bezoars that were identified in 35 patients (1 patient had a recurrent episode). The median age was 64 (range 30-94) years, with a male / female ratio of 27:9 (3:1). The majority of the study population was of Chinese ethnic origin (88.9%), which is 12.7% higher than the overall local Chinese population (76.2%). All of the patients presented with signs and symptoms of intestinal obstruction such as vomiting, abdominal pain and distention.
1. Risk factors
The medical history and risk factors are summarized in Table 1. There were 14 (38.9%) patients who had previous abdominal surgery. In particular, 6 (16.7%) had previous gastric surgery. Up to 20% of patients had documented usage of proton-pump inhibitors for gastritis/peptic ulcer disease. There were 4 patients (11.1%) who had a prior history of bezoars. Diabetes mellitus was the most common medical disease in the study population (44.4%). There were 6 (16.7.%) of the patients who were edentulous and were more likely to require enterotomy / bowel resection.
2. Imaging
Abdominal X-rays were performed initially for all patients, and showed dilated small bowel loops. All patients had a CT scan for further evaluation of SBO. CT scans showed evidence of, or suspected bezoars in the majority of cases (77.1%). In cases whereby CT scans were non-diagnostic for bezoars, these patients were statistically significantly more likely to eventually require enterotomy / bowel resection (88.9% vs 29.6%, p < 0.01).
3. Intra-operative findings and intervention
The median time from admission to operative intervention was 6.5 hours (range 1-120 hours). There were 20 (52.8%) cases that required only finger fragmentation and milking of the bezoar past the ileocaecal valve. The remaining 16 (44.4%) cases necessitated either an enterotomy (12 cases), or bowel resection (4 cases) for the removal of the bezoars.
The level of the obstructing bezoar is recorded in Table 1, with the majority of obstructed lesions found in the ileum (55.5%). Bezoars that were lodged in the ileum had significantly larger volumes than those in the jejunum (86.5 mL vs 63 mL, p = 0.04). In addition, bezoars lodged more proximally in the jejunum, were more likely to be fragmented and milked past the ileocaecal valve, while bezoars located more distally in the ileum were significantly more likely to require either an enterotomy or bowel resection (60% vs 25%, p = 0.01; Table 2). The need for enterotomy or bowel resection was also associated with a larger volume bezoar (mean 252 mL vs 68 mL, p < 0.01) and edentulous patients (31.3% vs 5%, p = 0.04).
All intra-operative cases were examined for synchronous bezoars, which were detected in 22.2% of all cases. The type of bezoars can only be determined in cases that underwent enterotomy / bowel resection. Of these cases, all bezoars were phytobezoars, with mushroom bezoars accounting for 37.4% of these cases. There was 1 case in which the bezoar was identified as a durian seed (a fruit unique to Southeast Asia that has a large hard seed) which the patient accidentally swallowed.
4. Morbidity and mortality
There was a higher rate of overall morbidity in the group that required enterotomy / bowel resection (43.8% vs 5%, p < 0.01; Table 1). There were 3 cases of wound infection, 3 cases of pneumonia, 1 case of anastomotic bleeding, and 1 case of sepsis with multi-organ failure. The need for enterotomy / bowel resection was also associated with a longer postoperative hospital stay (8 days vs 5 days). The overall mortality rate in the study population was 5.4% (2 cases).
Discussion
Predisposing risk factors for bezoar formation include previous gastric surgery, poor mastication, diabetes, and a high fiber diet. Gastric surgery has been thought to alter the stomach physiology in various ways that increase the risk of bezoar formation. The loss of pyloric function allows a large mass of undigested food to enter the small bowel resulting in impaction downstream. Truncal vagotomy (a traditionally common procedure to treat peptic ulcer disease), results in a hypoacidic environment, and reduced motility of the stomach [10]. Gastric acid-reduction medications such as proton-pump inhibitors can also alter the gastric pathology in a similar way to truncal vagotomy, and have also been associated with bezoar formation in patients with an intact gastro-intestinal tract [11,12].
Dietary habits also increase the risk of phytobezoar formation. Locally in Yishun Central, Singapore, the dried Shitake mushroom is considered a delicacy by the Chinese population, and regularly eaten, especially during festive periods. The stalk of the mushroom is particularly hard and fibrous, and patients who are edentulous, or have poor mastication function, will swallow the stalk as a complete piece [13] (Figures 1 and 2). Not surprisingly, there have been a corresponding increase in patients presenting with bezoar related intestinal obstruction during these festive periods, with 35% of these cases presenting within a 2-month window (January-February) in which the Chinese New Year usually falls (based on the lunar calendar). With an ageing population locally, there is an increasing number of elderly patients who may be edentulous, have poor dental hygiene, or poorly fitted dentures. These factors reduce mastication, resulting in reduced fiber break down, and an increased likelihood of forming bezoar in the gastro-intestinal tract. The results of this study indicate that the elderly population are more likely to have larger bezoars that will require enterostomy / bowel resection.

CT scan with axial and sagittal cuts with red arrows pointing to a non-specific intraluminal mass with upstream bowel dilatation.
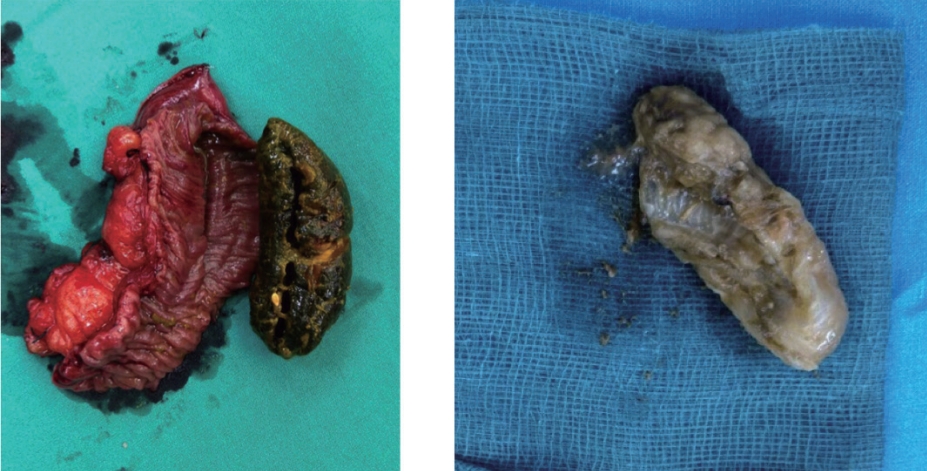
The case in Figure 1 above after small bowel resection (for possible small bowel tumor) showed that the intra-luminal mass was in fact a mushroom phytobezoar.
SBO from a bezoar remains a surgical emergency whereby prompt diagnosis and management is required to minimize morbidity and mortality. CT imaging has allowed clinicians to make quicker and more accurate diagnosis of bezoars, resulting in an earlier surgical intervention to remove the obstructing lesion. In this study, the median time from admission to operative intervention was 6.5 hours. The use of a CT scan can also provide information on the level of obstruction, and also detect the presence of multiple synchronous bezoars at other sites, and this can aid pre-operative planning. Typical radiological findings of small bowel bezoars on the CT image includes a well-defined round or ovoid mass and a heterogenous internal structure with a mottled gas appearance [15]. While a CT scan is superior compared with other imaging modalities in detecting bezoars, with a diagnostic rate of 77.1% in this study, there are also several pitfalls. There is a certain overlap of imaging features on the CT scan between bezoars and the small bowel “feces sign,” which is usually a longer segment of gas-containing feces-like material found proximal to the dilated small bowel [9]. This usually indicates a more chronic and insidious onset of SBO from any causes. A chronic long segment of small bowel bezoar can also resemble that of a small bowel “feces sign”, or they can also co-exist with the same presentation.
In this study, whilst the majority of cases were diagnosed pre-operatively using a CT scan (75%), there remained a minority of cases whereby imaging was non-diagnostic. These cases were usually associated with a larger volume of bezoar found intra-operatively, and also more likely to require enterotomy / bowel resection. In these cases of bezoars, there are usually long-segments which are more chronic in nature, and therefore may show a small bowel “feces sign,” instead of the typical bezoar features. In addition, mushroom phytobezoars are also more difficult to characterize by CT imaging as they do not always have the classic mottled gas appearance, and may instead resemble an intra-luminal tumor (Figures 1 and 2).
Whilst the laparoscopic approach has been described in the literature, this is only suitable for gastric bezoars [9,15]. The traditional open approach is recommended in cases of small bowel dilatation to reduce the risk if the bowel is injured. The most common site of impaction in this study was near the terminal ileum, which correlates with the narrowest point of the small bowel, approximately 60 cm from the ileocaecal valve. Impaction at the terminal ileum was observed in this study to be also associated with a larger volume of bezoar that necessitated either an enterotomy or bowel resection. Bowel ischemia is usually uncommon if there are no delays in presentation or diagnosis, and prompt treatment is initiated. Attempts should be first made to fragment the bezoar and milk it to the caecum, especially in cases of a smaller volume bezoar, reserving enterotomy and extraction in cases where this was not possible [16]. Fragmenting the bezoar and milking is associated with shorter post-operative stays, and lesser morbidity rates as observed in this study. However, forceful milking of bezoars can also lead to mucosal injury to the intestine, and clinical judgment should be made by an experienced surgeon if an enterotomy is necessary. Lastly, intra-operative assessments for synchronous bezoars are also recommended as they are present in 30%-40% of cases [17,18].
A large national audit has shown that complications such as superficial wound infection and dehiscence were more common in those patients that required small bowel resection than those without, following surgery for SBO [19]. This is also evident from observations in this study whereby larger bezoars were more likely to require enterotomy / bowel resection, and thus these patients were at higher risk of post-operative complications requiring longer periods of hospital stay.
Conclusion
CT imaging has made diagnosis of bezoars related SBO easier, allowing prompt intervention. However, there are still some pitfalls with CT imaging in identifying chronic large volume bezoars or mushroom phytobezoars. In this study, a more distally located obstruction in the ileum was associated with a larger volume bezoar requiring enterotomy / bowel resection. This was also associated with increased morbidity rates and a longer post-operative hospital stay.
This study has shown correlations between the risk factors associated with the need for enterotomy / bowel resection in SBO caused by bezoars, such as patients being edentulous, nondiagnostic CT scans, more distally located, and larger volume bezoars. These results can potentially allow optimal pre-operative counseling to patients regarding surgical options and post-operative care. While larger volume bezoars are observed to be more likely to require enterotomy / bowel resection, nevertheless possible fragmentation and milking should be attempted first intra-operatively, as it has lower morbidities. The need for enterotomy / bowel resection should be decided based on intra-operative findings, and the surgeon’s clinical judgment, as the bezoar needs to be completely removed whilst maintaining the integrity of the bowels.
Notes
The authors declare that there were no conflicts of interest or any grant support associated with this research.
Acknowledgements
We thank our institution clinical research unit biostatistician Dr Wang Jiexun, for her invaluable input.